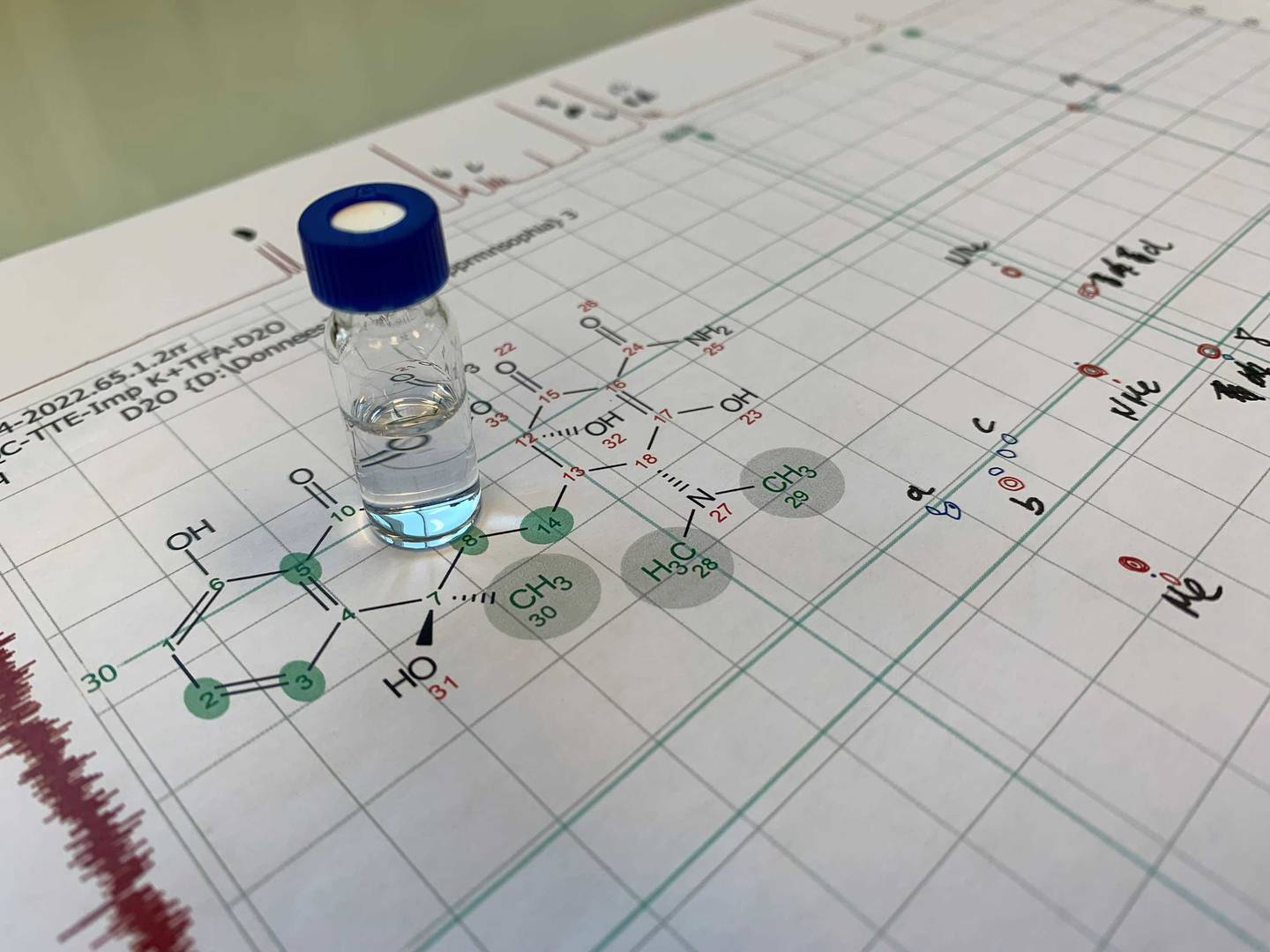
A complete [1H], [13C] and [15N] assignment of an unknown codeine impurity
The identification and structural elucidation of impurities is a routine requirement in process development according to the ICH Q3A(R2) and Q3B(R2) guidelines. Indeed, impurity identification is critical because impurities can affect drug safety, efficacy and quality. Identifying and controlling them helps ensure compliance with regulatory standards and protect patient health. In this case, an unknown impurity was found in a codeine drug product syrup during a yearly stability study. This raised safety concerns and required quick action to characterise the impurity and perform a toxicological assessment in order to exclude any patient risk.
High-resolution mass spectrometry (HRMS) investigations indicated an oxidation of codeine that had no instances of occurrence in known literature. Hydrogen peroxide oxidation of codeine was performed to enrich sample with the targeted impurity, submitted to preparative high-performance liquid chromatography (HPLC) and fully characterised by nuclear magnetic resonance (NMR) techniques. Position of oxidation and absolute configuration were determined using constant time inverse-detection gradient accordion rescaled heteronuclear multiple bond correlation (CIGAR-HMBC) spectroscopy at natural abundance.
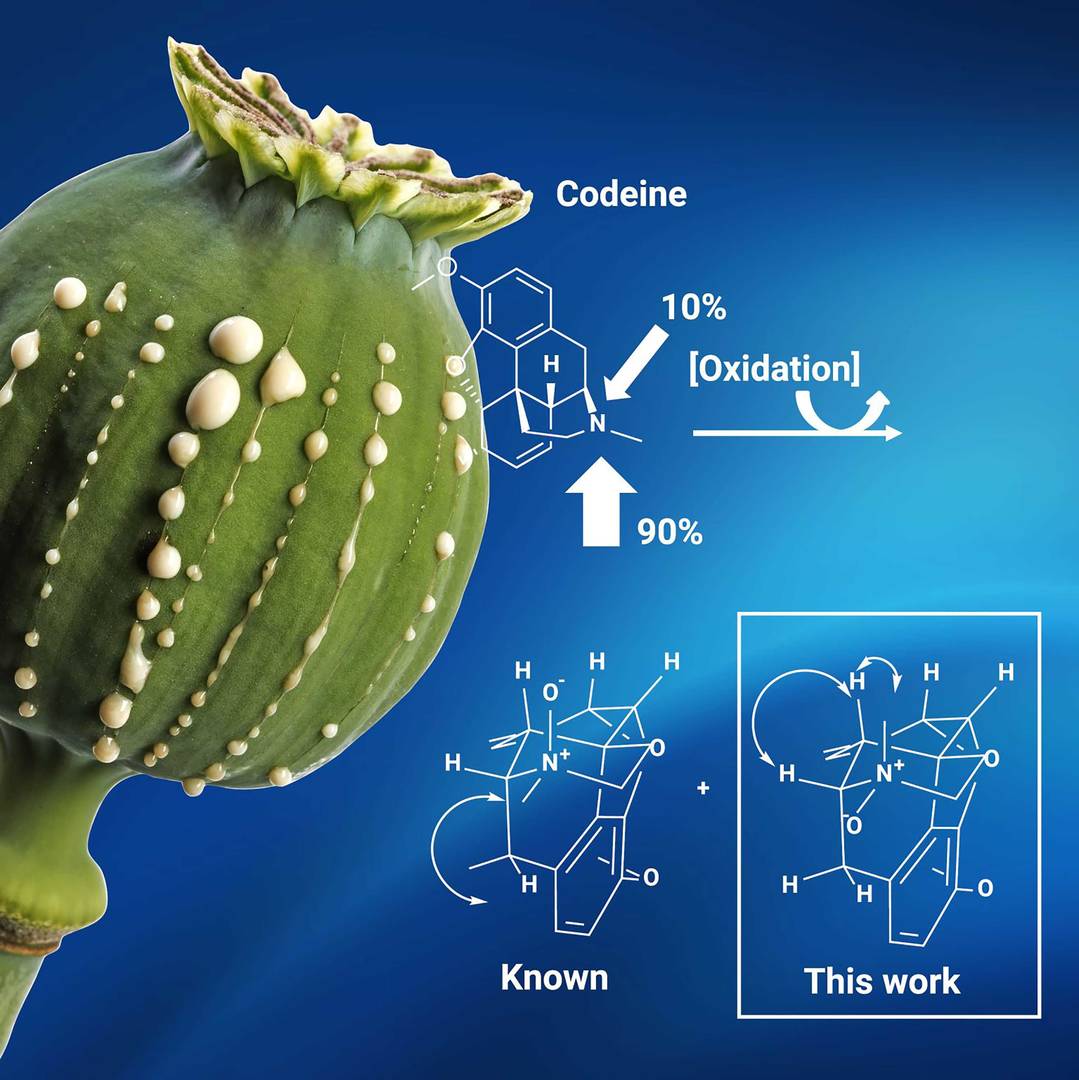
Elucidation of codeine and its (known and newly discovered) impurities

HRMS as an efficient and accurate impurity identification method
| Structural elucidation |
|
| Handling of controlled substances |
|
| Impurity isolation |
|
| Impurity synthesis |
|
| Characterisation |
|
Our highly experienced scientists can isolate, identify, synthesise and assess unknown impurities present at very low levels (<0.1 %) at every stage of the drug development lifecycle. Our chemists can identify degradation pathways and potential ingredient interactions that will facilitate the examination of dossiers by regulatory agencies.
Our capabilities include:
CONTACT US
Boiteau, J.-G. et al., Complete [1H], [13C] and [15N] assignments of the minor isomer of codeine N-oxide, Magn Reson Chem, 60, 12, 1185-1188 (2022).https://doi.org/10.1002/mrc.5302
Discover how our scientists isolate and characterise unknown impurities in drug substances and drug products.
Learn More
Nuvisan develops and manufactures high-potency APIs and controlled substances with full expertise.
Learn More
Discover our end-to-end services helping ensure safe, consistent and high-quality drug development in line with regulations.
Learn More