
Lead identification and optimisation using a combination of high-throughput screening and structure-based design
ATR kinase is a key mediator of DNA damage signalling and the replication stress response. Inhibition of ATR leads to generation of DNA double strand breaks, ultimately leading to tumour cell death. In the effort to advance cancer treatment, precision targeting of the DNA damage response (DDR) pathway is opening new possibilities for combating tumours. ATR kinase inhibitors, such as BAY 1895344 (Elimusertib), represent a promising step forward in exploring innovative therapeutic approaches.
As part of a project at a third-party company with which they were affiliated at the time, a team of Nuvisan scientists developed and preclinically characterised a potent, highly selective, orally available ATR inhibitor by a combined high-throughput screening and structure-based optimisation approach.

ATR inhibitor journey from hit to candidate BAY 1895344 using HTS, homology modelling and medicinal chemistry.
Utilising ultra-high throughput screening (uHTS) and structure-based drug design, BAY 1895344 was identified as a potent molecule with a low-nanomolar inhibitor profile. BAY 1895344 (Elimusertib) demonstrates significant efficacy as a monotherapy in preclinical cancer models, particularly in tumours with DDR deficiencies such as ATM mutations, and shows synergy when combined with DNA-damaging agents (e.g. Carboplatin) or DDR-compromising therapies (e.g. PARP inhibitors such as Olaparib). In xenograft studies, BAY 1895344 effectively reduced tumour growth in multiple cancer types, including ovarian, colorectal and prostate cancers. The combination of BAY 1895344 with antiandrogens therapy (Darolutamide) and external beam radiotherapy further enhanced therapeutic outcomes in hormone-dependent prostate cancer models (Lücking et al., J. Med. Chem (2020); Wengner et al., Mol. Canc. Ther. (2020)).
These findings support the potential of ATR inhibitors as versatile anticancer agents for personalised combination therapies targeting DNA repair vulnerabilities.

Wengner et al., Mol Cancer Thera 2020, Figure 6
Efficacy of BAY 1895344 (Elimusertib) in combination with Darolutamide in the hormone-dependent LAPC-4 human prostate cancer model
Clinical potential: BAY 1895344 (Elimusertib) was under clinical investigation for cancer with DDR defects, representing opportunities for trial support in oncology research.
Combination therapy synergy: Exploration of protocols that include ATR inhibitors alongside existing chemotherapies or radiotherapies for expanded treatment regimes.
Safety profiling: Tolerability insights from preclinical models highlight critical dose optimisation strategies for clinical applications.
Efficient drug discovery: BAY 1895344 (Elimusertib) exemplifies the use of uHTS to identify potent, selective cancer therapeutics targeting DDR pathways, enabling fast preclinical and clinical progress.
| Biochemical assays |
|
| Protein engineering and expression |
|
| High-throughput screening |
|
| Cell-based assays |
|
| In vivo studies |
|
| Chemical and analytical methods |
|
| Digital approaches |
|
Wengner, A. M. et al., The novel ATR inhibitor BAY 1895344 is efficacious as monotherapy and combined with DNA damage-inducing or repair-compromising therapies in preclinical cancer models. Mol Cancer Ther 19, 1, 26–38 (2020). (https://dx.doi.org/10.1158/1535-7163.MCT-19-0019)
Lücking, U. et al., Damage incorporated: discovery of the potent, highly selective, orally available ATR inhibitor BAY 1895344 with favourable pharmacokinetic properties and promising efficacy in monotherapy and in combination treatments in preclinical tumour models. J Med Chem 63, 13, 7293–7325 (2020). (https://dx.doi.org/10.1021/acs.jmedchem.0c00369)

Our comprehensive high-throughput screening services can support you with the identification of your hit molecules.
Learn More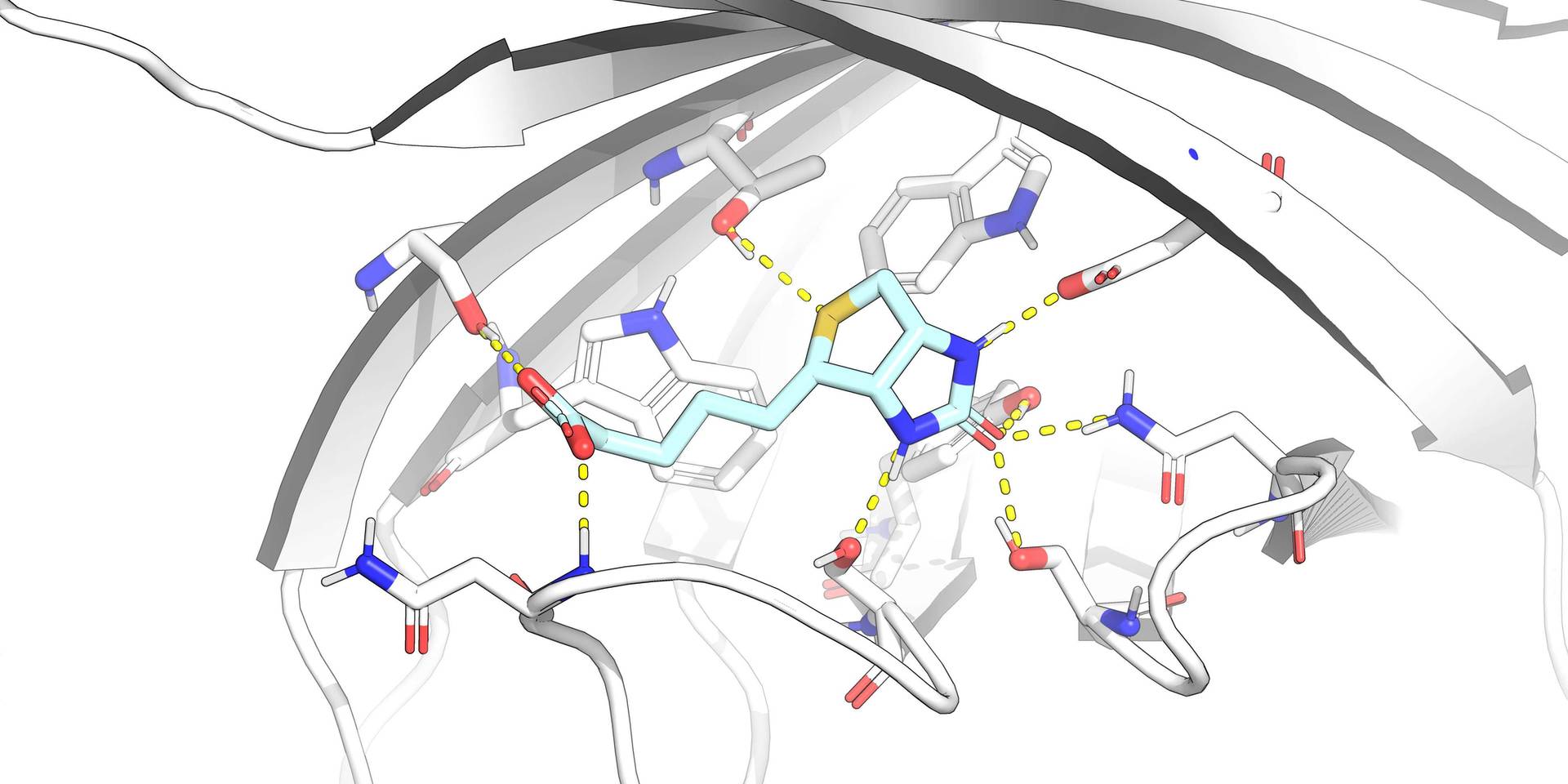
Leverage structural biology data though molecular modelling to accelerate access to improved chemical matter.
Learn More
Conducting reliable animal studies to help advance drug candidates towards clinical trials.
Learn More