
Our oncology services for drug discovery drive the advancement of cancer therapeutics through specialised expertise and cutting-edge technologies. We offer in vitro and in vivo models, biomarker discovery and PK/PD studies. By combining precision oncology with genomic insights, we help enhance candidate selection and improve clinical outcomes.
learn more
Our immuno-oncology services are dedicated to advancing breakthrough cancer immunotherapies through our expertise and state-of-the-art technologies. We offer in vitro and in vivo models, immune profiling, biomarker identification, checkpoint inhibitor research and combination therapy assessments. By harnessing precision immunology and genomic insights, we refine candidate selection and evaluate immune responses effectively.
Our metabolic disease services are dedicated to enhancing treatments for conditions like diabetes, obesity and metabolic syndrome using our specialized expertise and cutting-edge technologies. We provide in vitro and in vivo models, biomarker discovery, target validation and PK/PD studies. Through metabolic profiling and precision medicine strategies, we streamline candidate selection and accelerate therapeutic development.
learn more
Our inflammation and immunology services for drug discovery focus on developing therapies for autoimmune and inflammatory diseases using specialised expertise and cutting-edge technologies. We provide in vitro and in vivo models, biomarker discovery, immune cell profiling, cytokine analysis and target validation. By combining immunological insights with precision medicine, we enhance candidate selection and refine therapeutic strategies.
Nuvisan provides advanced research solutions to speed up the creation and development of treatments for various skin conditions, using cutting-edge models and analytical techniques.
learn more
Our neuroscience services for drug discovery are dedicated to developing therapies for neurological disorders using specialised expertise and advanced technologies. We provide in vitro and in vivo models, biomarker identification, behavioral assessments and target validation. By combining neurobiological insights with iPSC approaches, we enhance candidate selection and refine therapeutic strategies.
learn more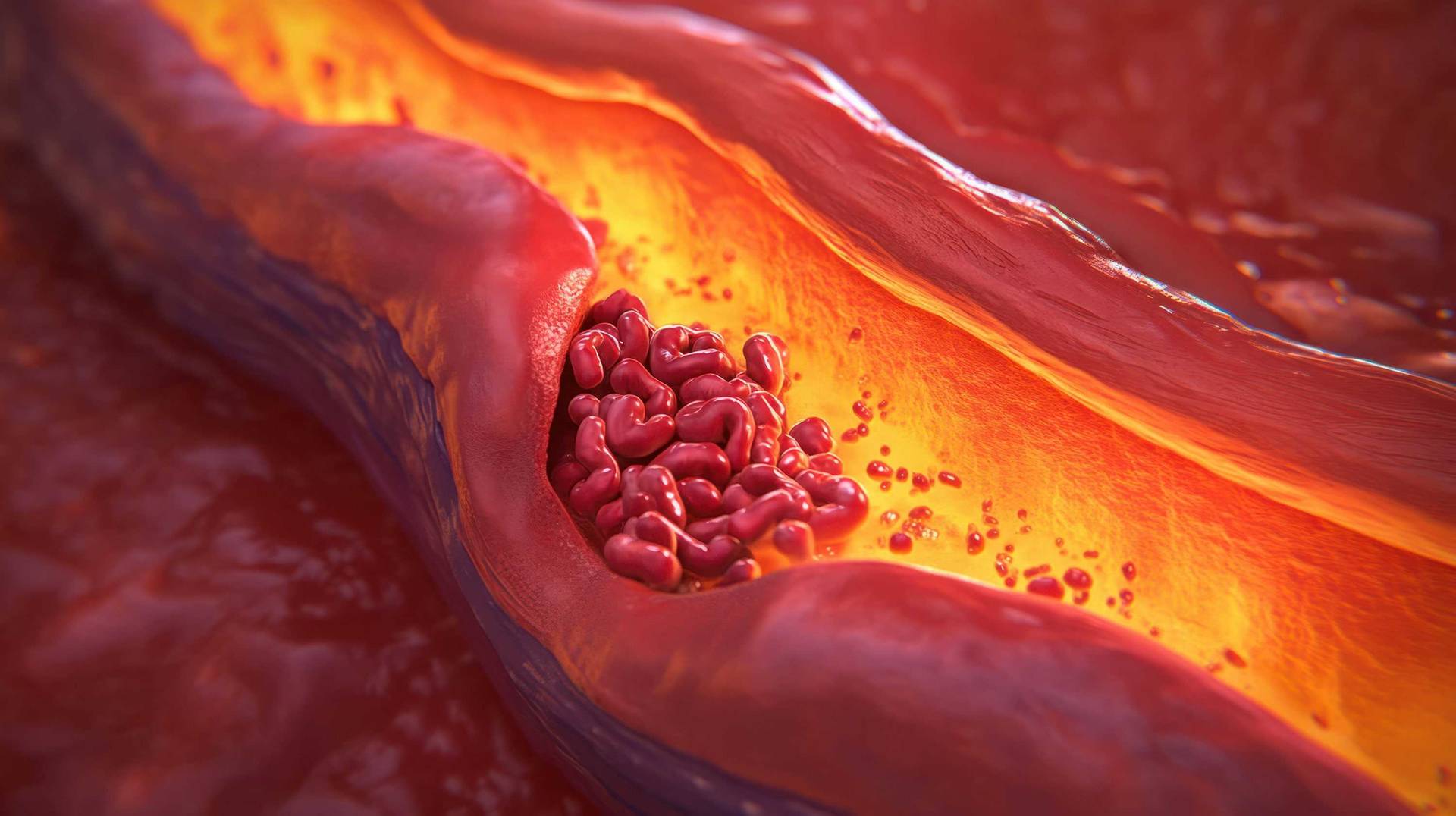
In the cardiovascular field, our experts focus on advancing treatments for heart and vascular disorders through specialised expertise and innovative technologies. We provide customised in vitro models, biomarker identification and target discovery to tackle the complexities of cardiovascular diseases. Integrating precision medicine, genomic insights and iPSC approaches enables us to optimise candidate selection and accelerate therapeutic development.
learn more
Our rare disease services for drug discovery are dedicated to developing therapies for conditions with low prevalence and significant unmet medical needs. We provide specialised in vitro (iPSCs) and in vivo models, biomarker discovery and genetic analysis of rare diseases. By utilising advanced genomic technologies and precision medicine, we can enhance candidate selection and therapeutic development.
learn more
Our male and female health solutions focus on addressing the unique health challenges of both men and women. We offer specialised research tools, including gender-specific disease models, biomarker discovery and therapeutic target validation. By integrating hormonal and genetic insights, we design personalised treatment approaches. With a strong emphasis on precision medicine, we collaborate with pharmaceutical and biotech partners to develop and advance innovative therapies tailored to men's and women's health needs.
learn more